
The strange mechanics of pregnancy in one Australian shark
People tend to think of sharks as large, frightening predators with sharp teeth, so it might come as a surprise to learn that some shark babies grow in the same way as humans – attached to the mother by an umbilical cord and placenta.
Our recent research published in the Journal of Comparative Physiology B sheds light on the placenta of the pint-sized Australian sharpnose shark – wherein a thin layer of egg capsule separates the mother’s and baby’s tissues.
Shark pregnancy
There are more than 500 different species of shark, some as small as your hand (like the dwarf lantern shark) and others as big as a bus (such as whale sharks).
Reproduction in sharks is equally varied: some lay eggs, but most give birth to live young. Sharks typically give birth after 11–12 months of pregnancy, but some, such as the frilled shark, are pregnant for more than three years.
In some sharks, a placenta develops during pregnancy. The placenta helps the baby shark breathe, eat and expel waste as it develops inside the mother. Other species of shark don’t have a placenta, and instead their babies feed on egg yolk, secretions, unfertilised eggs or even their own siblings.
Biologists have long been fascinated by reproductive diversity in sharks, but haven’t yet figured out why some sharks have placentas and others don’t.
The Australian model for shark pregnancy
In Australia, we have a unique model to understand shark pregnancy: the Australian sharpnose shark (Rhizoprionodon taylori). This small shark is just 70 centimetres long, and lives off the coast of northern Australia.

An adult Australian sharpnose shark is just 70cm long. CSIRO National Fish Collection
The females get pregnant every summer with five to ten babies (called pups). Shortly after falling pregnant, embryonic development is paused foseven months. During this time (called diapause), the embryos are still just a bundle of cells.
Diapause ensures the babies are born in summer, almost a year later. This is when they stand the best chance of surviving, as water temperature is at its highest and food is abundant.
Diapause ends when signals, such as hormones trigger the embryos to resume development. They feed on egg yolk at first, and the placenta takes over about a month later. Placentas connect to the babies by umbilical cords, and transport enough nutrients to allow them to grow 300 times larger over just 4.5 months of development.
When the babies are born, their umbilical cords drop off, leaving them with belly buttons. They can almost immediately hunt and fend for themselves, which is astonishing considering humans are born helpless after nine months of pregnancy.

The diapause period in the Australian sharpnose shark lasts about seven months.
Our recent studies looked at how the placenta supports sharpnose babies so effectively during pregnancy.
The unique features of shark placentas
Under the microscope, we discovered the sharpnose placenta is made up of thin layers of cells from the mother and the baby, separated by an extremely thin egg capsule (0.00005 centimetres). Structurally, this is very similar to the placentas of most other sharks.
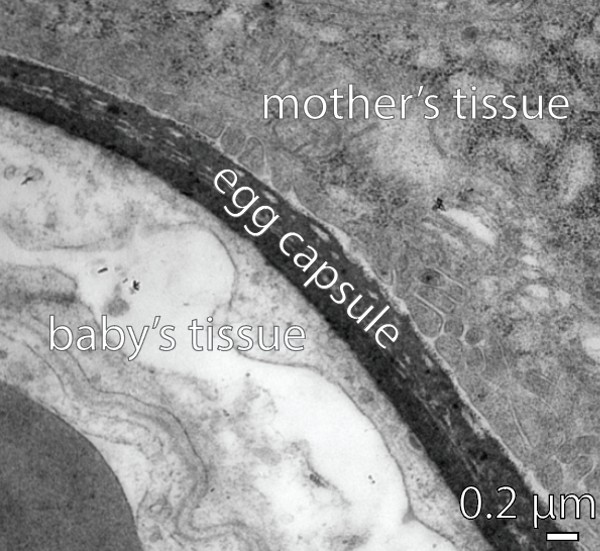
In Rhizoprionodon taylori mothers, the placenta has a thin egg capsule separating the mother’s and baby’s tissues.
The egg capsule in the sharpnose placenta has no pores. But we showed it can still allow small molecules to pass from the mother to her babies, such as oxygen and small nutrients including sugars, amino acids, fatty acids and water. This explains how sharpnose babies can grow so quickly inside the mother’s uterus.
However, the size of the molecule matters – and larger proteins can’t get through the egg capsule. We think the capsule may act as a physical barrier that protects the babies from bacteria in the uterus during pregnancy.
Physically separating the mother’s and baby’s genetically different tissues could also help stop the mother’s immune system from attacking the baby.
Human placentas look completely different to shark placentas – there is no egg capsule and the baby’s tissues are directly bathed in their mother’s nutrient-rich blood.
Yet both function to nourish babies during pregnancy. This similar function is an amazing example of convergent evolution, given humans and sharks are separated by 450 million years of evolution.
Further research into this little Australian shark will help us understand how placentas that evolved independently in different species can perform the same functions during pregnancy.
This article was first published on The Conversation and written by Dr Camilla Whittington and Alice Buddle from University of Sydney and Professor Colin Simpfendorfer from James Cook University.