
Researchers find surprising twist behind rare neurological disorder
A fruit fly study, by an international group of researchers, has pinpointed how a genetic mutation causes one of the important molecular machines in our cells to malfunction. The findings provide a biological mechanism behind episodic ataxia 6, a rare neurological disorder.
Episodic ataxia 6 is a debilitating illness that severely impacts balance and coordination and causes vertigo. Patients with episodic ataxia 6 are known to have mutations in the gene SLC1A3, that codes for glutamate transporters. Glutamate transporters are molecular machines on the surface of our cells that control chemical signals and make sure our cells seamlessly talk to one another, in particular our neurons, and are vital for normal brain function.
The importance of glutamate transporters in nerve signalling and memory is widely acknowledged, but even what they look like in our cells has remined a mystery until recently.

Visual representation of a glutamate transporter
A previous University of Sydney study found the glutamate transporter not only looks like a ‘twisted elevator’, but is also an efficient multi-tasker, transporting glutamate, water and chloride ions at the same time.
Until now, the exact impact of the SLC1A3 mutations had on glutamate transporters was unknown.
In this study, researchers found the SLC1A3 mutation triggered a ‘fault’ in the ‘twisted elevator’ mechanism, in particular the ability of the glutamate transporter to move chloride ions.
The mutation didn’t just increase or increase chloride flow, the error went both ways.
The researchers found fruit fly larvae with the genetic mutation that impacted chloride flow walked round in circles, and their movements were unsteady.
But in an unexpected twist to the story, the researchers found that the mutations only impacted chloride ion transport. There was no change to the ability to move glutamate.
The results could help our understanding of the condition and in the future develop treatments for patients suffering from episodic ataxia 6 and similar disorders.
“A key step towards improved treatment for episodic ataxia 6 is knowing whether the disease stems from an issue in glutamate transport, or the chloride channel, or even both,” said senior author Professor Renae Ryan from the School of Medical Sciences, Faculty of Medicine and Health.
“Our results strongly support that the mutations impact the chloride channel.”
The discovery, published in The Journal of Clinical Investigation, was co-led by researchers from the University of Sydney, and the Research Institute of the McGill University Health Centre, in collaboration with University of Illinois computational scientists.
Mapping the mutations in fruit flies
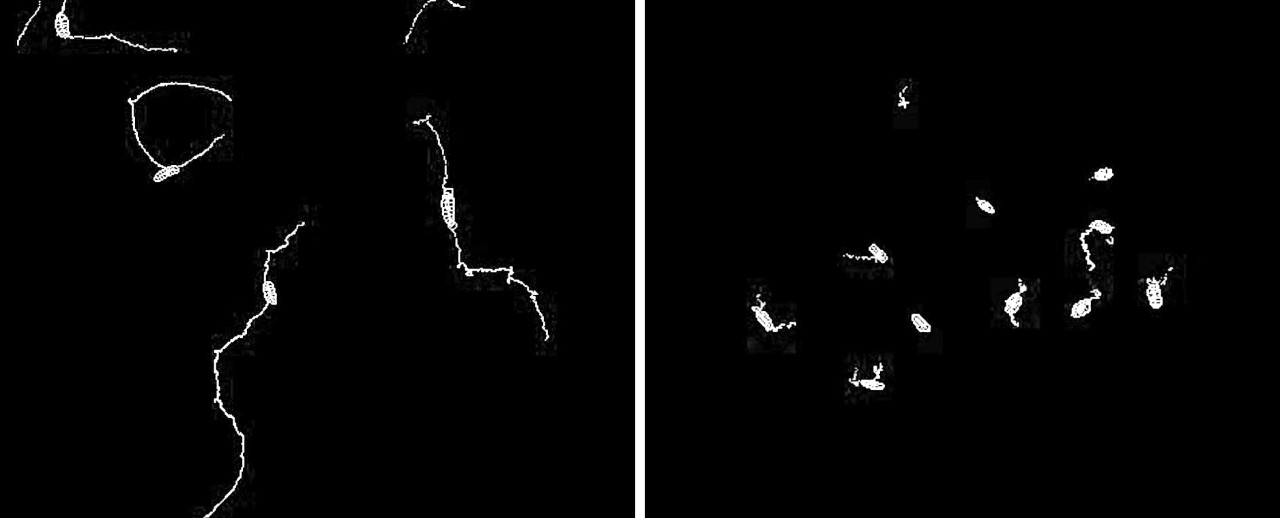
The researchers tracked the movement of fruit flies to see the impact of the mutations (white lines). Fruit fly larvae with the genetic mutation that affected chloride flow moved around unsteadily in the same place (right), compared to fruit fly larvae without the mutation which were able to move around normally (left). (Wu et al.)
“Studying the role of glutamate transporter protein mutations in episodic ataxia 6 is not straightforward. This protein has a dual function as both a glutamate transporter and as a chloride channel.” Said Professor Don van Meyel, senior scientist in Brain Repair and Integrative Neuroscience Program at McGill University.
“The chloride channel function is activated by the glutamate transport function, making it difficult to disentangle one function from the other.”
The research team carefully analysed the impact of five sub types of episodic ataxia 6 related mutations.
“We studied the impacts of these mutations in frog oocytes (eggs), then using the fruit fly model, we demonstrated that an increase or decrease in glutamate transporter chloride channel activity can affect motor function,” said first author Qianyi Wu, PhD, who was a PhD student with Professor Ryan at the time of this study.
“We were able to show that several mutations in glutamate transporter impacted the chloride channel while the glutamate transporter function was unaffected.”
“This suggests that episodic ataxia type 6 may be directly linked to how chloride levels are balanced in the brain, and not to glutamate transport,” adds co- author Azman Akhter who was a Masters of Science student with Professor van Meyel.
“Since the glutamate transporter is highly concentrated in glial cells in the human brain, we believe they contribute to chloride homeostasis, an idea supported by our fruit fly model.”
“Additionally, we were able to identify a mechanism by which one severe mutation in the SLC1A3 gene also causes an unusual leak of sodium ions through the glutamate,” explains Dr Shashank Pant, who conducted computer modelling of the glutamate transporter structure in the lab of collaborator Dr Emad Tajkhorshid at the University of Illinois.
Science in action
This study is an example of how scientific knowledge about the inner workings of our cells are built up over years of research. Professor Ryan says each study along the way has been crucial to gradually building their understanding.
“This is a really nice translational study moving from basic protein characterisation and computer modeling to examining its role and what happens during disease in an animal model.”
The researchers involved in the study were: Qianyi Wu and Renae M. Ryan from the from the School of Medical Sciences, Faculty of Medicine and Health, University of Sydney. Azman Akhter, Donald J. van Meyel, Alastair Garner and Tomoko Ohyama from McGill University, Shashank Pant and Emad Tajkhorshid from the University of Illinois at Urbana-Champaign, Chicago. Eunjoo Cho and Jin Xin Zhu from the Research Institute of the McGill University Health Centre.
Declaration: The study was approved by the University of Sydney Animal Ethics under the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes.