
Spinal cord stimulation doesn't help with back pain, says new review
Spinal cord stimulation, a medical technology suggested to treat people with chronic back pain, does not provide long-term relief and may cause harm, according to a Cochrane Review released today.
Spinal cord stimulation is thought to work by implanting a device that sends electrical pulses to the spinal cord to interrupt nerve signals before they get to the brain.
The study reviewed published clinical data on spinal cord stimulation. This included randomised controlled trials, considered to be the most robust method to measure effectiveness of a treatment in medical research.
The researchers analysed the results of 13 clinical trials, looking at data from 699 participants, comparing spinal cord stimulation treatment with placebo or no treatment for low back pain.
Cochrane reviews are trusted by researchers, medical professionals and policymakers because they use robust methodologies to combine evidence from multiple sources, reducing the impact of bias and random error that can make individual studies less reliable.
The review concluded that spinal cord stimulation is no better than a placebo for treating low back pain, with probably little to no benefit for people with low back pain or improvement in their quality of life.
There was little to no clinical data regarding the long-term effectiveness of spinal cord stimulation.
Our review of the clinical data suggests no sustained benefits to the surgery outweigh the costs and risks.
The researchers also found that adverse side effects to the surgery were poorly documented overall, preventing them from concluding the level of risk involved. Harms from spinal cord stimulation could include nerve damage, infection, and the electrical leads moving, all of which may need repeated surgeries.
The review findings have been submitted to the Federal Department of Health and Aged Care prosthesis list review taskforce. The taskforce is reviewing the eligibility of current prostheses subsidised by Medicare.
In Australia, the devices' long-term safety and performance are also being re-assessed by The Therapeutic Goods Administration (TGA), the country’s regulatory authority for therapeutic goods.
“Spinal cord stimulation is invasive and has a great financial cost to people who choose surgery as a last resort to alleviate their pain. Our review found that the long-term benefits and harms are essentially unknown,” said lead researcher Dr Adrian Traeger from Sydney Musculoskeletal Health, an initiative of the University of Sydney, Sydney Local Health District and Northern Sydney Local Health District.
“Our review of the clinical data suggests no sustained benefits to the surgery outweigh the costs and risks.
“Low back pain is one of the leading causes of disability worldwide. Our findings further emphasise the urgent need to review funding arrangements for chronic pain care to help patients in their search for relief. There are evidence-based physical and psychological therapies for back pain; ensuring access to these is essential.”
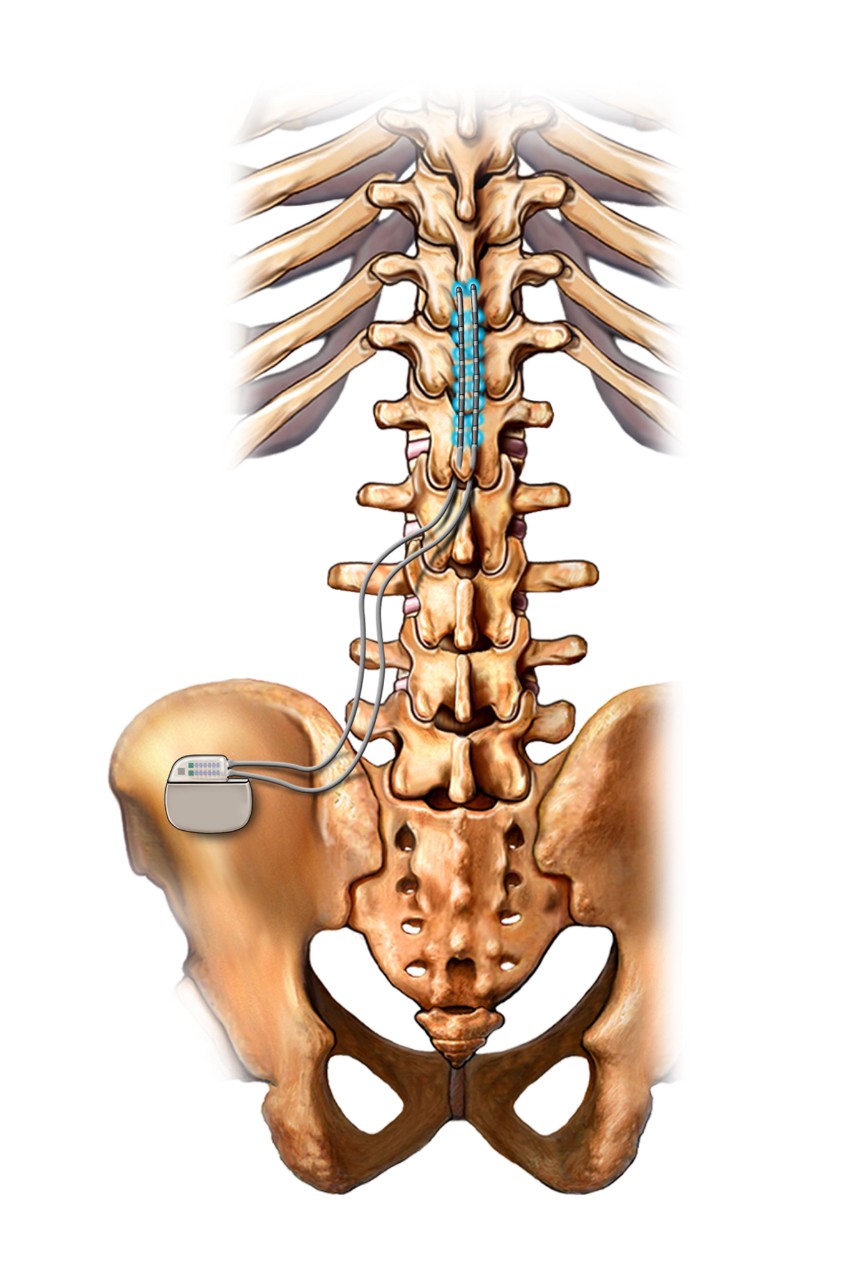
Spinal cord simulator device. Image credit: Alamy
The review team found multiple gaps in clinical data.
There were no studies that investigated the long-term (more than 12 months) impact of spinal cord stimulation on low back pain. The longest was a single six-month trial.
The majority of clinical trials only looked at the immediate impact of the device, which is a time frame of less than a month.
The review team provided a list of recommendations, including that future spinal cord stimulation clinical trials be at least 12 months, clearly document the number of people who experience adverse events and make comparisons with other pain treatment options.
Professor Chris Maher, Co-Director of Sydney Musculoskeletal Health, said:
“Our review found that the clinical benefit of adding spinal cord stimulation to treat low back pain remains unknown. When coupled with the reality that these devices are very expensive and often break down there is clearly a problem here that should be of concern to regulators.”
A separate Cochrane review, in which the researchers were not involved, examined the effect of spinal cord stimulation versus placebo in people with chronic pain. Similar to this review, it concluded there was a lack of evidence to suggest long-term benefits in treating chronic pain.
Declaration: The researchers declare no conflicts of interest.
Image: Adobe Stock