
The bionic eye that could help blind people navigate the world
For people with impaired eyesight or no sight at all, the world is an enormous obstacle course. Professor Gregg Suaning is pushing vision capture and nerve stimulation technologies beyond their limits to help clear a path.
As research goals go, it is bold with perhaps a touch of the miraculous. For more than twenty years, Professor Gregg Suaning has been working to bring sight to the blind. Though in the early days, the idea was almost all he had.
“We were dismantling radios and car electricals to make the equipment we needed,” Suaning says, obviously enjoying the memory. “One time we were making bespoke electrodes and we ended up using a capacitor out of a big, old‑style television.”
Those early hurdles have long been cleared and today, Suaning’s work is at a point where the broad technology of delivering a sense of sight exists. In principle, it’s not unlike the cochlear implant and related technologies which now help millions of hearing‑impaired people.
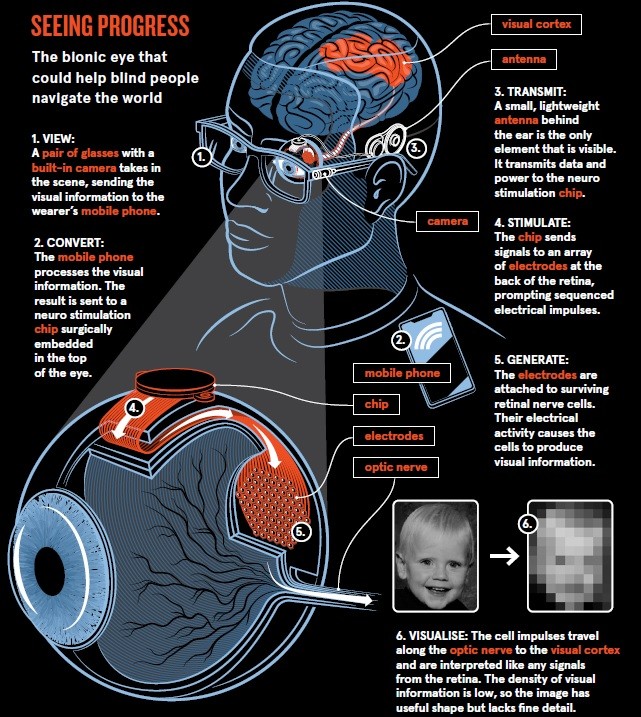
A camera on a pair of glasses collects the visual information which is then sent to a mobile phone and processed. The result is sent wirelessly to a microchip implanted in the retina which decodes the wireless signal and sends electrical impulses to the part of the brain that produces vision: the visual cortex.
While the technology might be cochlear-like, the degree of difficulty is many times greater because vision is so much more information dense than sound.
Where hearing technologies can deliver a more than acceptable result using 14 channels of information, Suaning’s work currently uses 100 channels with more limits in the process of being pushed.
Still, the challenge plays to Suaning’s natural impulses since he grew up wanting to be a motor mechanic before discovering mechanical engineering then biomedical engineering. In fact, he was an engineer at Cochlear in its very early days, and it was there that he set himself the goal of helping blind people see.
“It hasn’t been easy,” says Suaning in his Australian-inflected San Franciscan accent, having met his Australian wife in a Jerusalem Youth Hostel.
“Keeping in mind that full vision is like a million channels of information, it really helps that the brain can also make a lot out of very little.”
This was demonstrated in 2014 when the sight technology was still cumbersome, and lab bound. As part of national project where researchers were developing an Australian bionic eye, three blind volunteers came to a Melbourne University lab, and were implanted with a rudimentary electrode array and connected to laboratory-based electronics.
“Two didn’t get much of a reaction, but one of them did really well,” remembers Suaning. “Going through an obstacle course she avoided and even identified obstacles, including a chair. Though it wouldn’t have been a fully realised chair, just a few dots. But her brain filled in enough of the gaps.”
This represents a key challenge for the research: making the visual information captured and communicated actually meaningful for the blind person, meaningful being a key term here.
“We’re working towards something so blind people can navigate the world,” Suaning says. “To help them recognise objects, avoid obstacles and move about more confidently. Will it ever be the full visual experience sighted people have? Thinking about the huge advances there’ve been in video game technology, it might be possible, but we’re not there yet.”
Still, over the years people have been inspired by the huge promise of the technology and even offered much needed financial support (“One blind person called me wanting to donate to the work - though I think he was deaf as well. He was absolutely screaming into the phone because he was so excited”.)
But recent support from the Neil and Norma Hill Foundation has sent Suaning and the technology down a new path.
Where the research focus for Suaning has always been on a rare, blindness inducing condition called retina pigmentosa, which can strike people in their 30s and 40s, causing initial tunnel vision that later narrows to full vision loss, the Foundation gift brought macular degeneration into the frame.
As the Trustee for the Foundation says, “When we heard about Professor Suaning's work we felt compelled to help. We are so pleased that our philanthropy could help to ease the burden in some way for people with macular degeneration and their families."
In some ways, MD is like the reverse of retina pigmentosa since the damage starts in the middle of the field of vision and works outward. Certainly, it’s much more common than retina pigmentosa, with one in seven Australians over 50 having some age-related macular degeneration (MD), and about 17% of those going on to suffer vision loss.
That being the case, the reason Suaning didn’t focus on MD in the early days of his research was out of concern for his volunteers. People with severe retina pigmentosa can suffer full vision loss, whereas people with MD usually retain some sight around the central area of retinal damage. Suaning didn’t want to risk whatever sight was still retained by MD volunteers.
Allowing that significant advances have been made since those early days, Suaning is now preparing for the first human trials where the volunteers will use wearable equipment that they can take home. To even contemplate using human volunteers in this way, Suaning has had to demonstrate the safety of what he’s doing to the highest possible standards.
This he has done, and in the process seen for himself that the newest expression of the technology would present a minimal threat to any volunteer participating, including someone with macular degeneration.
This has also seen him looking differently at the technology itself. For retina pigmentosa, the in-eye electrode array is arranged for the outside-to-inside progress of the condition, whereas the new MD array must work inside-to-outside.
Having to do this thinking has fed new and useful ideas into the process.
Another thing that has helped Suaning’s work is his move to the University of Sydney, “There’s so much multidisciplinary stuff that happens here,” he says. “As part of one ecosystem there’s the technical side, medical, business, even the psychological aspects.
“That makes every advancement even more immediate. You can see the effect it could have, maybe not tomorrow, but you can see it; something is emerging that will help people.”